
Accordingly, investors should monitor Pardes’ Investors website, in addition to following Pardes’ press releases, Securities and Exchange Commission filings, public conference calls, presentations and webcasts. Pardes intends to use the Investors page of its website ( ) as a means of disclosing material non-public information and for complying with its disclosure obligations under Regulation FD (Fair Disclosure).

For more information, please visit Availability of Other Information about Pardes Biosciences PBI-0451 is currently in a Phase 1 placebo-controlled, blinded, randomized, dose escalation study in healthy volunteers in New Zealand evaluating the safety, tolerability, and pharmacokinetics of PBI-0451 after single and multiple ascending doses. PBI-0451 is being developed for the treatment and prevention of SARS-CoV-2 infection and associated diseases. This protease is highly similar across all coronaviruses, including known and emerging coronavirus variants. PBI-0451 is an investigational orally bioavailable direct-acting antiviral (DAA) inhibitor of the main protease (Mpro), an essential protein required for the replication of coronaviruses, including the novel SARS-CoV-2 that causes COVID-19. For more information, please visit About PBI-0451 We are applying modern reversible-covalent chemistry as a starting point to discover and develop novel oral drug candidates, including our lead product candidate PBI-0451, while reimagining the patient journey to access these medicines. Pardes Biosciences is a clinical-stage biopharmaceutical company created to help solve pandemic-sized problems, starting with COVID-19. Pardes anticipates reporting data from this ongoing study at a scientific conference later this quarter.
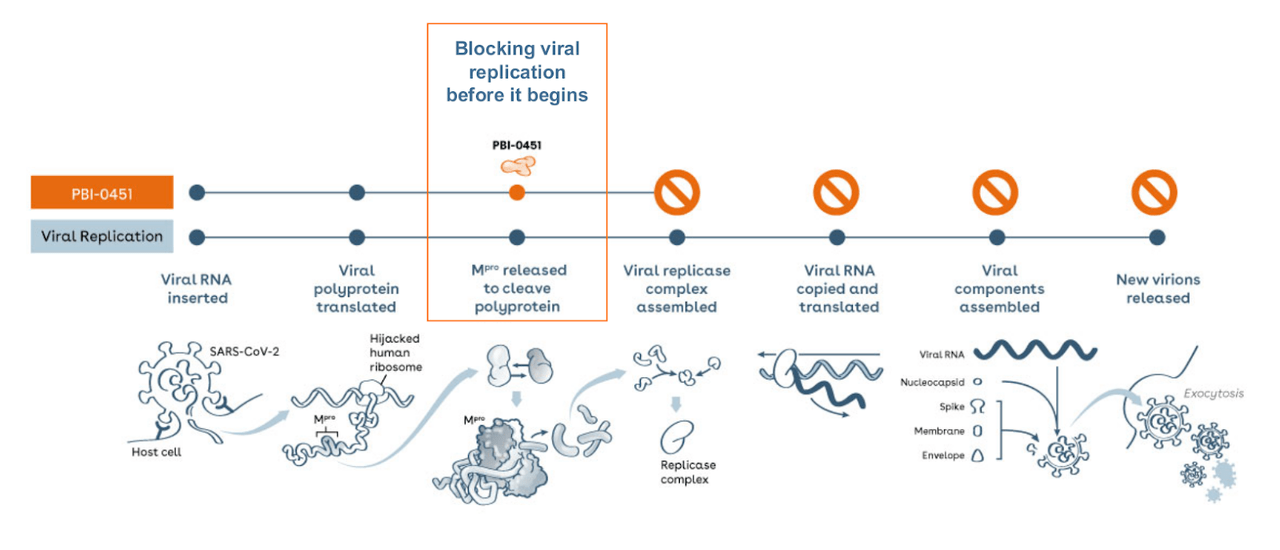
PB-0451 is currently under evaluation in a Phase 1 placebo-controlled, blinded, randomized, dose escalation study in healthy volunteers in New Zealand evaluating the safety, tolerability, and pharmacokinetics of PBI-0451 after single and multiple ascending doses. Pending additional engagement with FDA and other regulators, we anticipate initiating our global Phase 2/3 studies of PBI-0451 in SARS-CoV-2 infected patients in mid-2022.” “The clearance of our IND for PBI-0451 enables us to proceed with the initiation of additional Phase 1 clinical trials for PBI-0451 in the U.S. “As we enter our third year of a global pandemic, we believe the value of oral direct-acting antivirals for SARS-CoV-2 has become increasingly clear, especially if they can be given as standalone therapies,” said Uri A. (NASDAQ: PRDS), a clinical-stage biopharmaceutical company developing PBI-0451 as a potential novel direct-acting, oral antiviral drug candidate for the treatment and prevention of SARS-CoV-2 infections and associated diseases (i.e., COVID-19), today announced that its Investigational New Drug (IND) application for PBI-0451 has been cleared by the United States Food and Drug Administration (FDA). 03, 2022 (GLOBE NEWSWIRE) - Pardes Biosciences, Inc.
